7 results
Integrated efficacy analysis from phase 3 studies of investigational microbiome therapeutic, SER-109, in recurrent Clostridioides difficile infection
- Matthew Sims, Michael Silverman, Thomas Louie, Elaine Wang, Colleen Kraft, Mayur Ramesh, Tatiana Bogdanovich, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Christine Lee, Paul Feuerstadt, Darrell Pardi, Colleen Kelly, Peter Daley, Godson Oguchi, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
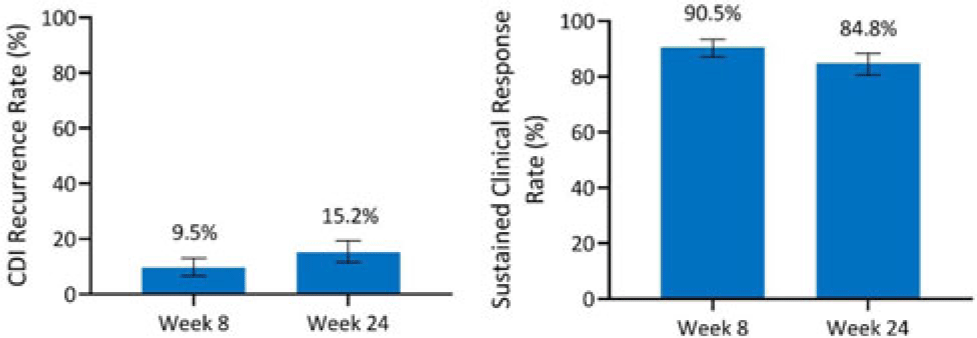
Background: Antibiotics alone are often insufficient to treat recurrent C. difficile infection (rCDI) because they have no activity against C. difficile spores that germinate within a disrupted microbiome. SER-109, an investigational, oral, microbiome therapeutic comprised of purified Firmicutes spores, was designed to reduce rCDI through microbiome repair. We report an integrated efficacy analysis through week 24 for SER-109 from phase 3 studies, ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a randomized, placebo-controlled phase 3 trial conducted at 56 US or Canadian sites that included 182 participants with ≥2 CDI recurrences, confirmed via toxin EIA testing. Participants were stratified by age (<65 years or ≥65 years) and antibiotic regimen (vancomycin, fidaxomicin) and were randomized 1:1 to placebo or SER-109 groups. ECOSPOR IV was an open-label, single-arm study conducted at 72 US or Canadian sites including 263 participants with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III who experienced on-study recurrence diagnosed by toxin EIA (n = 29) and (2) participants with ≥1 CDI recurrence (diagnosed by PCR or toxin EIA), inclusive of the current episode (n = 234). In both studies, the investigational product was administered orally as 4 capsules over 3 consecutive days following symptom resolution after standard-of-care antibiotics. The primary efficacy end point was rCDI (recurrent toxin-positive diarrhea requiring treatment) through week 8. Other end points included CDI recurrence rates and safety through 24 weeks. Results: These 349 participants received at least 1 dose of SER-109 in ECOSPOR III or ECOSPOR IV (mean age 64.2; 68.8% female). Overall, 77 participants (22.1%) enrolled with their first CDI recurrence. Four participants received blinded SER-109 in ECOSPOR III followed by a second dose of open-label SER-109 in ECOSPOR IV. Overall, the proportion of participants who received any dose of SER-109 with rCDI at week 8 was 9.5% (33 of 349; 95% CI, 6.6 %–13.0%), and the CDI recurrence rate remained low through 24 weeks (15.2%, 53 of 349; 95% CI, 11.6%–19.4%), corresponding to sustained clinical response rates of 90.5% (95% CI, 87.0%–93.4%) and 84.8% (95% CI, 80.6%–88.4%), respectively (Fig. 1). Most rollover participants (25 of 29, 86.2%) were from the placebo arm; 13.8% had rCDI by week 8. Conclusions: In this integrated analysis, the rates of rCDI were low and durable in participants who received the investigational microbiome therapeutic SER-109, with sustained clinical response rates of 90.5% and 84.8% at weeks 8 and 24, respectively. These data further support the potential benefit of microbiome repair with SER-109 following antibiotics for rCDI to prevent recurrence in high-risk patients.
Financial support: This study was funded by Seres Therapeutics.
Disclosure: None
Integrated safety analysis of phase 3 studies for investigational microbiome therapeutic, SER-109, in recurrent CDI
- Matthew Sims, Charles Berenson, Stuart Cohen, Elaine Wang, Elizabeth Hohmann, Richard Nathan, Alberto Odio, Paul Cook, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Bret Lashner, Louis Korman, Doria Grimard, Juan Carlos Moises Gutierrez, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s44-s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) often recurs in patients aged ≥65 years and those with comorbidities. Clinical trials often exclude patients with history of immunosuppression, malignancy, renal insufficiency, or other comorbidities. In a phase 3 trial (ECOSPOR III), SER-109 was superior to placebo in reducing recurrent CDI (rCDI) risk at week 8 and was well tolerated. We report integrated safety data for SER-109 in a broad patient population through week 24 from phase 3 studies: ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a double-blind, placebo-controlled trial conducted in participants with ≥2 CDI recurrences randomized 1:1 to placebo or SER-109. ECOSPOR IV was an open-label, single-arm study conducted in 263 patients with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III with on-study recurrence and (2) participants with ≥1 CDI recurrence, inclusive of the current episode. In both studies, the investigational product was administered as 4 oral capsules over 3 days. Treatment-emergent adverse events (TEAEs) were collected through week 8; serious TEAEs and TEAEs of special interest (ie, bacteremia, abscess, meningitis) were collected through week 24. Results: In total, 349 participants received SER-109 in ECOSPOR III and/or ECOSPOR IV (mean age 64.2; 68.8% female). Chronic diseases included cardiac disease (31.2%), immunocompromised or immunosuppressed (21.2%), diabetes (18.9% ), and renal impairment or failure (13.2%). Overall, 221 (63.3%) of 349 participants who received SER-109 experienced TEAEs through week 24. Most were mild to moderate and gastrointestinal. The most common (>5% of participants) treatment related TEAEs were flatulence, abdominal pain and distension, decreased appetite, constipation, nausea, fatigue, and diarrhea. No participants experienced a treatment-related TEAE leading to study withdrawal. Invasive infections were observed in 28 participants (8%); those with identified pathogens were unrelated to SER-109 species, and all were deemed unrelated to treatment by the investigators. There were 11 deaths (3.2%) and 48 participants (13.8%) with serious TEAEs, none of which were deemed treatment related. There were no clinically important differences in the safety profile across subgroups of sex, race, prior antibiotic regimen, or number of CDI recurrences. No safety signals were observed in participants with renal impairment or failure, diabetes, cardiac disease, or immunocompromised or immunosuppressed individuals. Conclusions: In this integrated analysis of phase 3 trials, SER-109, an investigational microbiome therapeutic, was well tolerated in this vulnerable patient population with prevalent comorbidities. No infections, nor those with identified pathogens, were attributed to SER-109 or product species. This safety profile might be expected because this purified product is composed of spore-forming Firmicutes normally abundant in the healthy microbiome.
Financial support: This study was funded by Seres Therapeutics.
Disclosures: None
Healthcare resource utilization in a phase 3 trial of SER-109 in patients with recurrent Clostridioides difficile infection
- Stuart Cohen, Thomas Louie, Charles Berenson, Alpesh Amin, David Lombardi, Sissi Pham, Shirley Huang, Elaine Wang, Brooke Hasson, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s73
-
- Article
-
- You have access Access
- Open access
- Export citation
-
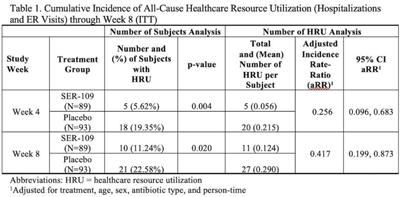
Background: The estimated economic cost of Clostridioides difficile infection (CDI) is $5.4 billion annually, primarily attributed to acute-care costs. We previously reported data from ECOSPOR III that SER-109, an investigational oral microbiome therapeutic, was superior to placebo in reducing recurrent CDI (rCDI) in adults at 8 weeks after treatment, with a 68% relative risk reduction. Adults with rCDI have more hospitalizations and emergency room (ER) visits (defined herein as healthcare resource utilization, HRU) compared to those without recurrence. Thus, we evaluated incidence of HRU. Methods: Adults with rCDI (≥3 episodes in 12 months) were screened at 56 US and Canadian sites and were randomized 1:1 to SER-109 (4 capsules × 3 days) or placebo following resolution of CDI with standard-of-care CDI antibiotics. The primary end point was rCDI at 8 weeks. Exploratory end points included cumulative incidence of hospitalizations through 24 weeks after treatment. Here, we report cumulative incidence of all-cause HRU through 8 weeks after treatment. Results: In total, 281 patients were screened and 182 were randomized (59.9% female; mean age 65.5 years; 98.9% outpatient). Overall, 31 patients (17%) had 38 hospitalizations or ER visits through week 8 (11 events in 10 SER-109 patients and 27 events in 21 placebo patients) (Table 1). The cumulative incidence of HRU was lower in SER-109–treated patients compared to placebo at both weeks 4 and 8 with most events (65.8%) recorded within 4 weeks after treatment. The adjusted HRU incidence rate (by person time, age, sex, and antibiotic use) was also lower in SER-109–treated patients compared to placebo at weeks 4 and 8 (0.256 [95% CI, 0.096–0.683] versus 0.417 [95% CI, 0.199–0.873], respectively). Conclusions: SER-109–treated patients had less HRU compared to placebo patients through 8 weeks after treatment in this mostly outpatient population. These data suggest a potential benefit of SER-109 in reducing HRU, thus lowering the healthcare burden of rCDI.
Funding: Seres Therapeutics
Disclosures: None
Efficacy and Safety of Investigational Microbiome Drug SER-109 for Treatment of Recurrent Clostridioides difficile Infection
- Barbara McGovern, Mathew Sims, Colleen Kraft, Elaine Wang, Kelly Brady, Christopher Ford, O’Brien Edward, Mary-Jane Lombardo, Jennifer Wortman, Kevin Litcofsky, Jennifer Mahoney, Christopher McChalicher, Jonathan Winkler, Sarah Garant, John Aunins, Matthew Henn, Lisa von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
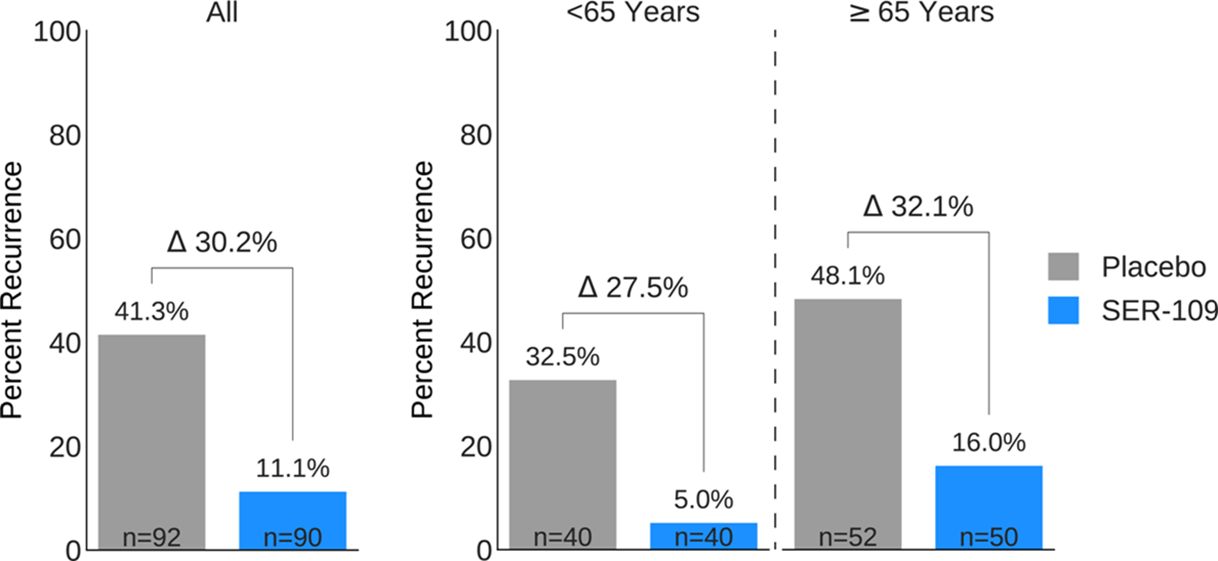
Background: Antibiotics targeted against Clostridioides difficile bacteria are necessary, but insufficient, to achieve a durable clinical response because they have no effect on C. difficile spores that germinate within a disrupted microbiome. ECOSPOR-III evaluated SER-109, an investigational, biologically derived microbiome therapeutic of purified Firmicute spores for treatment of rCDI. Herein, we present the interim analysis in the ITT population at 8 and 12 weeks. Methods: Adults ≥18 years with rCDI (≥3 episodes in 12 months) were screened at 75 US and CAN sites. CDI was defined as ≥3 unformed stools per day for <48 hours with a positive C. difficile assay. After completion of 10–21 days of vancomycin or fidaxomicin, adults with symptom resolution were randomized 1:1 to SER-109 (4 capsules × 3 days) or matching placebo and stratified by age (≥ or <65 years) and antibiotic received. Primary objectives were safety and efficacy at 8 weeks. Primary efficacy endpoint was rCDI (recurrent toxin+ diarrhea requiring treatment); secondary endpoints included efficacy at 12 weeks after dosing. Results: Overall, 287 participants were screened and 182 were randomized (59.9% female; mean age, 65.5 years). The most common reason for screen failure was a negative C. difficile toxin assay. A significantly lower proportion of SER-109 participants had rCDI after dosing compared to placebo at week 8 (11.1% vs 41.3%, respectively; relative risk [RR], 0.27; 95% confidence interval [CI], 0.15–0.51; p-value <0.001). Efficacy rates were significantly higher with SER-109 vs placebo in both stratified age groups (Figure 1). SER-109 was well-tolerated with a safety profile similar to placebo. The most common treatment-emergent adverse events (TEAEs) were gastrointestinal and were mainly mild to moderate. No serious TEAEs, infections, deaths, or drug discontinuations were deemed related to study drug. Conclusions: SER-109, an oral live microbiome therapeutic, achieved high rates of sustained clinical response with a favorable safety profile. By enriching for Firmicute spores, SER-109 achieves high efficacy while mitigating risk of transmitting infectious agents, beyond donor screening alone. SER-109 represents a major paradigm shift in the clinical management of patients with recurrent CDI. Clinicaltrials.gov Identifier NCT03183128. These data were previously presented as a late breaker at American College of Gastroenterology 2020.
Funding: Seres Therapeutics
Disclosures: None
Figure 1.
143 A Combination of Olanzapine and Samidorphan Has No Clinically Relevant Effect on QT Prolongation up to Supratherapeutic Doses
- Lei Sun, Sergey Yagoda, Hongqi Xue, Randy Brown, Narinder Nangia, David McDonnell, Bhaskar Rege, Lisa von Moltke, Borje Darpo
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 24 April 2020, p. 291
-
- Article
-
- You have access Access
- Export citation
-
Background:
ALKS 3831, a combination of olanzapine and samidorphan (OLZ/SAM) in development for schizophrenia, is intended to mitigate olanzapine-associated weight gain. This thorough QT (tQT) study evaluated OLZ/SAM effects on electrocardiogram parameters.
Methods:In this randomized, double-blind, parallel-group study, 100 patients with stable schizophrenia were randomized 3:2 to either receive OLZ/SAM 10/10 mg (therapeutic dose) on days 2–4, 20/20 mg on days 5–8, and 30/30 mg (supratherapeutic dose) on days 9–13 with moxifloxacin-matching placebo on days 1 and 14, or a single dose of moxifloxacin 400 mg and matching placebo on days 1 and 14 (nested crossover design). Drug concentration relation to change from baseline in Fridericia-corrected QTc (ΔQTcF) was evaluated using a linear mixed-effect concentration-QTc (C-QTc) model. Adverse events were assessed.
Results:The slope (90% CI) of the C-QTc was not significant for olanzapine or samidorphan (0.03 [−0.01, 0.08] and 0.01 [−0.01, 0.04] msec per ng/mL, respectively). Predicted placebo-corrected ΔQTcF (90% CI) was 2.33 (−2.72, 7.38) and 1.38 (−3.37, 6.12) msec at the observed geometric mean maximal concentration of olanzapine (62.6 ng/mL) and samidorphan (75.1 ng/mL), respectively, on day 13. A clinically relevant QT effect (ie, placebo-corrected ΔQTcF ≥10 msec) can be excluded for olanzapine and samidorphan concentrations up to ≈110 and ≈160 ng/mL, respectively. Assay sensitivity was confirmed by the C-QTc relationship of moxifloxacin. OLZ/SAM was well tolerated.
Conclusions:OLZ/SAM, in doses and plasma concentrations up to supratherapeutic levels, was well tolerated and had no clinically relevant effects on electrocardiogram parameters, including QT interval, in patients with schizophrenia.
Funding Acknowledgements:This study was funded by Alkermes, Inc.
Size matters: the importance of particle size in a newly developed injectable formulation for the treatment of schizophrenia
- Rakesh Jain, Jonathan Meyer, Angela Wehr, Bhaskar Rege, Lisa von Moltke, Peter J. Weiden
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 3 / June 2020
- Published online by Cambridge University Press:
- 21 May 2019, pp. 323-330
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
One of the challenges with initiating long-acting injectable (LAI) antipsychotic regimens is achieving relevant drug levels quickly. After first injection of the LAI antipsychotic aripiprazole lauroxil (AL), the lag to reaching relevant plasma aripiprazole levels was initially addressed using supplemental oral aripiprazole for 21 days. A 1-day AL initiation regimen using a NanoCrystal® Dispersion formulation of AL (ALNCD; Aristada Initio®) combined with a single 30 mg dose of oral aripiprazole has been developed as an alternative approach. We compared the 1-day AL initiation regimen (ALNCD + 30 mg oral aripiprazole for 1 day) with the 21-day AL initiation regimen (AL + 15 mg/day of oral aripiprazole for 21 days) using kinetic modeling. Observed and modeled data demonstrate that the 1-day AL initiation regimen provides continuous aripiprazole exposure comparable to the 21-day AL initiation regimen. Each component of the 1-day AL initiation regimen (30 mg oral aripiprazole, ALNCD, and AL) contributes to aripiprazole plasma levels at different times, with oral aripiprazole predominating in the first week, then ALNCD and AL over time. In a double-blind, placebo-controlled, phase 1 study in patients with schizophrenia, the 1-day initiation regimen resulted in rapid achievement of relevant plasma aripiprazole levels comparable to those from the 21-day initiation regimen. Safety and tolerability of the 1-day regimen were consistent with the known profile of aripiprazole. Each part of the 1-day initiation regimen, together with AL, is necessary for continuous aripiprazole exposure from treatment initiation until the next regularly scheduled AL injection is administered.
27 A New Method for Initiating Treatment with the Long-acting Antipsychotic Aripiprazole Lauroxil
- Jonathan Meyer, Rakesh Jain, Angela Wehr, Bhaskar Rege, Lisa von Moltke, Peter J Weiden
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, pp. 188-189
-
- Article
-
- You have access Access
- Export citation
-
STUDY OBJECTIVE
Slow release is a fundamental feature of long-acting injectable (LAI) antipsychotics. This property allows continuous drug exposure between dosing intervals. However, there can be a significant delay between giving the first LAI dose and achievement of efficacious plasma concentrations. This time period requires additional pharmacologic intervention. Until now, this delay was addressed with one of two strategies: 1) continuing with supplemental oral antipsychotic, or 2) giving more LAI up front (e.g. loading dose). A third strategy has now been developed to reduce the time needed for oral supplementation when starting the LAI aripiprazole lauroxil (AL) from 21days to 1day. A nano-crystalline milled dispersion of AL (ALNCD; brand name ARISTADA INITIO™) was formulated by reducing the AL particle diameter from micron-size particles to nanometer- sized particles. ALNCD has faster dissolution and a shorter half-life than AL and is designed to be used as a single injection along with a single oral aripiprazole dose of 30mg as a 1-day alternative to the 21days of oral aripiprazole supplementation. Here we provide an overview of the new 1-day initiation regimen for starting AL treatment, and demonstrate the relative contributions of each of its components.
METHODSA blinded, randomized, phase 1, pharmacokinetic (PK), and safety study compared the 1-day initiation regimen with the 21-day oral aripiprazole regimen. A combination of observed data, and population pharmacokinetic model–based simulations were used to plot plasma aripiprazole concentrations of single doses of ALNCD, 30mg oral aripiprazole, and AL, individually, and all three combined.
RESULTSThe PK profiles of the 1-day and 21-day initiation regimens (both in conjunction with either 441mg or 882mg doses of AL) were comparable, with therapeutically relevant aripiprazole levels achieved within 4days of treatment initiation. The safety profile of the 1-day initiation regimen was similar to the 21-day initiation regimen, and consistent with that of AL. Aripiprazole concentration–time profiles demonstrated that each component delivered aripiprazole to the systemic circulation at different time periods, with the 30mg dose of oral aripiprazole predominant in the first week, followed by ALNCD, and then AL.
CONCLUSIONSThe 1-day initiation regimen is well-tolerated and a suitable alternative to 21days of oral aripiprazole supplementation for starting AL. Each component of the 1-day initiation regimen, together with AL, is necessary to provide continuous coverage from treatment initiation until the next regularly scheduled AL injection.
Funding Acknowledgements: This study was funded by Alkermes Inc.






